CO2 Capture System Creates Usable Power
 Jan-23-19
A new carbon capture system inspired by the ocean turns CO2 into a useable power source.
Jan-23-19
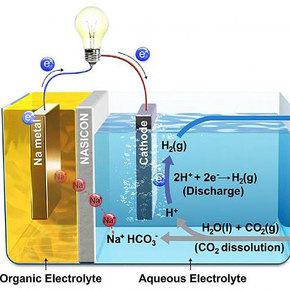
A new carbon capture system inspired by the ocean turns CO2 into a useable power source.Developed by teams from UNIST and Georgia Tech, the Hybrid Na-CO2 System is based on the knowledge that carbon dioxide can be dissolved in water to create an acidic solution. With that phenomenon to guide them, the team created a fuel cell made up of a sodium metal anode submerged in an organic electrolyte, and a cathode in an aqueous solution. Injecting CO2 into the electrolyte caused a reaction with the cathode that increased the acidity of the solution, generating electricity while also creating hydrogen. The system boasts a 50 percent CO2 conversion rate and, unlike conventional designs, releases the remaining CO2 as baking soda rather than back into the atmosphere as CO2 gas.
More Info about this Invention:
[CO.UK][NEWS.UNIST.AC.KR]

Add Your Comment