Methods for Diagnosis of Clostridium Diffcile and Methods and Vectors for Recombinant Toxin Expression
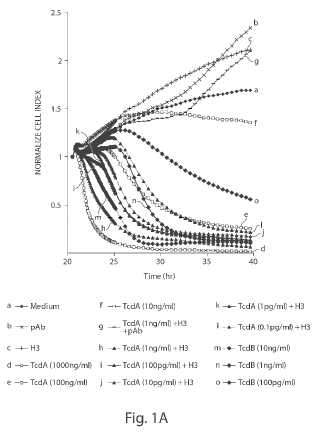
Cell-based methods for rapid real time assay of a presence of Clostridium difficile toxin and/or cells are provided, using an assay having a toxin-enhancing antibody and a sensitive cell line carrying Fc?R receptors, and kits for this assay. An ultrasensitive cell- based immunocytotoxicity assay for detecting less then 1 pg/ml of C. difficile toxins in clinical samples. The assay is simple, has a turnaround time of approximately 3 hours, and detects less than about 1 pg/ml of toxin A. The presence of C. difficile toxins in the fecal and serum specimens of experimentally infected piglets was detected with this assay. The cellular effects of TcdA were substantially enhanced via an opsonizing antibody through Fc gamma receptor I (Fc?RI)-mediated endocytosis.; A TcdA-specific monoclonal antibody, A1H3, was found to significantly enhance the cytotoxicity of TcdA to macrophages and monocytes. The A 1H3 -dependent enhancement of glucosyltransferase activity, cytoskeleton disruption, and TNF-a production induced by TcdA was demonstrated also in RAW 264.7 cells. The interaction of Fc?RI with A1H3 underlay the antibody-dependent enhancement of cellular effects of TcdA. Expression of Fc?RI in CHO cells strikingly enhanced their sensitivity to TcdA complexed with A1H3. Presence of A1H3 facilitated the cell surface recruitment of TcdA, conxributing to the antibody-dependent, Fc?RI -mediated enhancement of TcdA activity. The effect of chlorpromazine and endosomal acidification inhibitors indicated an important role of the endocytic pathway in AlH3-dependent enhancement of TcdA activity.; Methods for high level recombinant expression of C. difficile toxins in Bacillus cells, and vectors for expression, strains of Bacillus carrying the vectors are provided.Attached files:
Patents:
EP 2,288,719
Inventor(s): FENG HANPING [US]; TZIPORI SAUL [US]; YANG GUILIN [CN]
Type of Offer: Licensing
« More Diagnostic Patents
