Co-registration of PET and Transrectal Ultrasounds (TRUS) Images for Improved Prostate Imaging and Biopsy
APPLICATIONS OF TECHNOLOGY:Detects prostate cancer using accurately co-registered positron emission tomography (PET) and transrectal ultrasound (TRUS) images
Guides prostate biopsy sampling
Monitors response to treatment and detects local recurrence
ADVANTAGES:
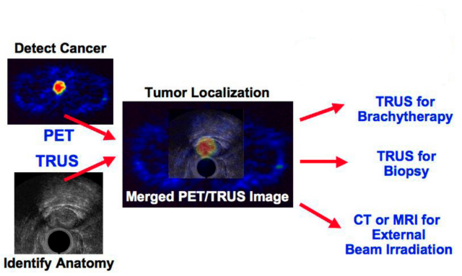
Provides high-resolution anatomical detail in the prostate region through TRUS images, allowing tumors detected by PET to be localized precisely
Compact and open prostate-optimized PET design allows convenient access for biopsy and therapeutic delivery
Significant cost savings, using only 25 percent of the detectors of a conventional full-body PET scanner
Higher signal-to-noise ratio than conventional PET scanners, due to compact elliptical shape and improved shielding
TRUS provides better prostate anatomical image detail, delineation and volume calculation than Computed Tomography
Transfer to commercial production presents no significant complications
ABSTRACT:
More than half of all malignant prostate tumors detected today cannot be directly palpated by a physician. Prostate-specific antigen and digital rectal exam screenings give high false-positive rates, and the diagnostic accuracy of biopsy is problematic. New imaging technology is needed to confirm diagnosis, guide biopsy, and help guide and monitor treatment.
Jennifer Huber, William Moses, Qiyu Peng, Ronald Huesman and Thomas Budinger at Lawrence Berkeley National Laboratory have devised a new dual-imaging technology for sensitive detection of prostate cancer, using a high-performance positron emission tomograph (PET) optimized to image the prostate, in concert with a commercial transrectal ultrasound system (TRUS).
Images are taken sequentially with both devices during a single patient session of roughly an hour. Functional imaging with PET detects malignant tumors in the prostate region and determines a tumor’s aggressiveness, and ultrasound imaging with a transrectal probe provides high-resolution volumetric anatomical detail.
The system includes a PET scanner with a patient bed, on which a transrectal ultrasound probe-linear stepper-arm assembly is mounted. The TRUS assembly moves to allow correct positioning of the probe in a patient, then its position is fixed and 3D TRUS data are acquired. While the patient and probe remain in the same position, the patient bed is then moved and two PET data sets are acquired. First, 511 keV PET point sources are attached to the TRUS stepper and are quickly imaged in the PET scanner. Second, the patient’s prostate is centered in the PET scanner and patient imaging is performed using a PET radiopharmaceutical such as [11C]choline. After the patient study, the point source data are used to determine the 3D location of the TRUS probe tip relative to the center of the PET scanner, so the PET and TRUS patient image sets can be simply and accurately co-registered. The PET and TRUS patient data are reconstructed in three dimensions and fused to show both anatomical and functional detail.
Given improved targeting with PET-TRUS image-directed cores, the diagnostic accuracy of prostate biopsy could be improved. And used in conjunction with a new generation of PET radiopharmaceuticals now undergoing clinical trials, PET-TRUS prostate imaging could provide a more accurate and speedy method to assess treatment response and detect local prostate cancer recurrence.
A custom PET-TRUS prostate phantom is used to validate and calibrate the dual imaging system. With structures that simulate the acoustical properties for TRUS and 511 keV activity concentrations for PET, the phantom is composed of agar-gelatin-based tissue-mimicking materials mixed with radioactive water solutions. It can be rendered compatible with computer tomography and magnetic resonance imaging.
REFERENCE NUMBER: IB-2420
Attached files:
Inventor(s): Jennifer Huber, William Moses, Qiyu Peng, Ronald Huesman and Thomas Budinger
Type of Offer: Licensing
« More Medical Devices Patents
« More Biomedical Patents
« More Medical Patents
