Mouse Models of Rhinovirus-induced disease
BackgroundRhinoviruses cause the common cold, hospitalization of infants, pneumonia in the immunosuppressed and the majority of acute exacerbations of asthma and chronic obstructive pulmonary disease (COPD). These diseases generate hundreds of billions of dollars in healthcare costs, and the morbidity and mortality attributable to rhinovirus infections is enormous. No effective treatment or vaccine is available.
Attempts to develop small-animal models of rhinovirus infection in many species, from mice to monkeys, have failed. The lack of small-animal models of rhinovirus infection has thus severely hampered mechanistic insight into and therapeutic development for rhinovirus infections over the past 50 years.
Ten percent of the ~100 rhinovirus serotypes make up the minor receptor group of viruses, which can use both the human and mouse forms of the receptor to enter cells of either species. The remaining 90% comprise the major group and use human ICAM-1 for cell attachment and entry, but these viruses do not bind mouse ICAM-1.
Invention
Researchers developed mouse models of major- and minor-group rhinovirus infection and of rhinovirus-induced asthma exacerbation. For the major group rhinovirus infection model transgenic mice expressing human ICAM-1 are used.
Applications
The mouse rhinovirus infection models enables scientists to:
• Study rhinoviruses and their mechanisms of disease development in infection alone and exacerbation of airways diseases
• Identify targets for the development of treatments for rhinovirus infection (currently non-existent)
Patent Status
A patent application has been filed by the owners and the technology is now available for license.
Type of offer: Licensing
Image Caption:
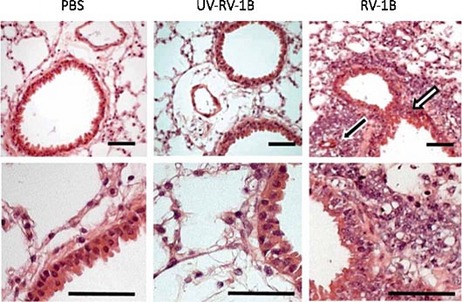
Live minor-group rhinovirus-1B, but not UV-inactivated rhinovirus-1B induces airway and lung inflammation and mucin production in BALB/c mice. Mice were mock infected (PBS) or infected with 5x10⁶ TCID₅₀ of rhinovirus-1B (RV-1B) or dosed with UV-inactivated rhinovirus-1B (UV–RV-1B). This picture represents H&E-stained lung sections taken at day 2 after treatment. Perivascular (black arrow) and peribronchial (white arrow) inflammation are indicated. Scale bars, 50 μm.
Attached files:
Type of Offer: Licensing
« More Medical Patents
